- Philips receives 510(k) clearance from the US Food and Drug Administration (FDA) to market its innovative EPIQ ultrasound system in the US
- Clinicians can now visualize deeper levels of anatomical detail and contrast resolution on patients of all types
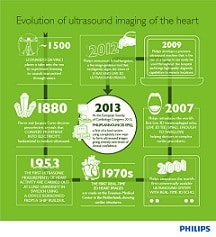
Amsterdam, Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA) today announced the launch of EPIQ ultrasound system, offering an entirely new level of clinical confidence with a first-of-its-kind ultrasound architecture that is a totally new approach to creating ultrasound images without compromise. Making its debut at the European Society of Cardiology (ESC) 2013 Congress in Amsterdam, EPIQ features a new imaging technology called nSIGHT that when combined with Philips’ Anatomical Intelligence technology, delivers a powerful combination of speed and image clarity.