June 25, 2018
Philips showcases ongoing commitment to the future of nuclear medicine at the 2018 SNMMI Annual Meeting
Portfolio includes unique solutions that enhance the patient experience and support diagnostic confidence
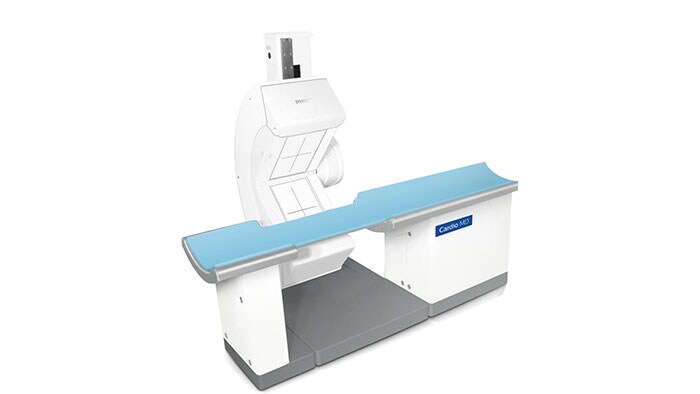
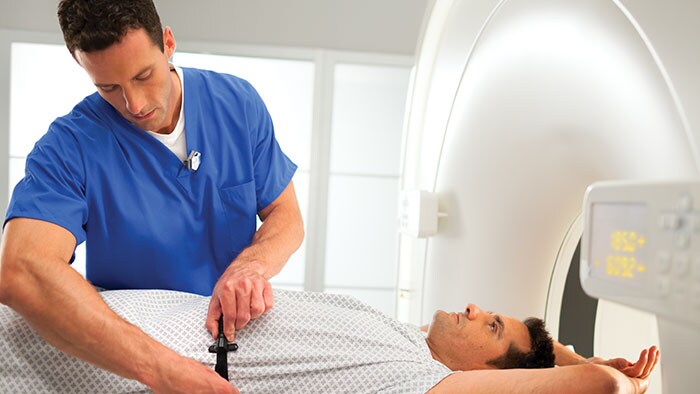
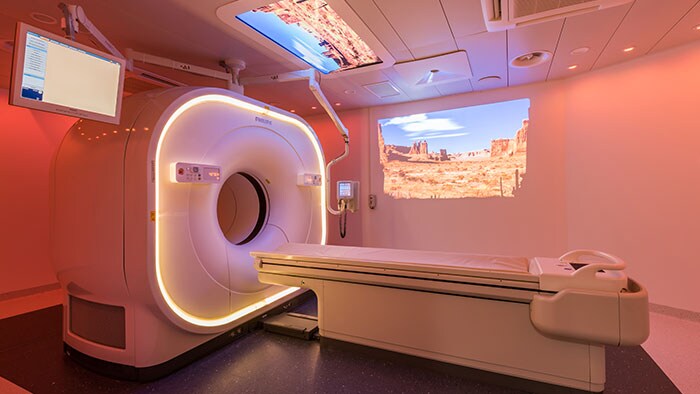
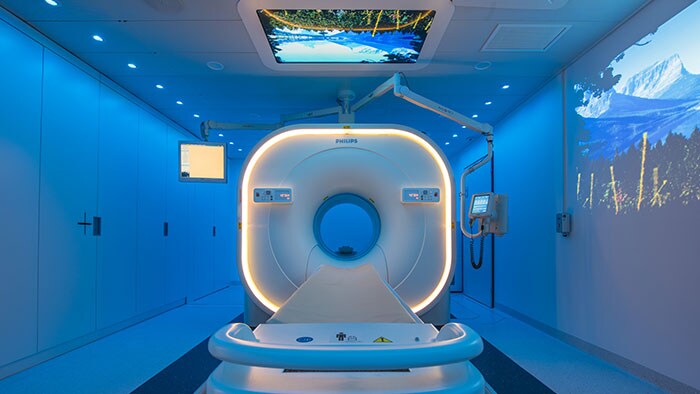
Amsterdam, the Netherlands and Philadelphia, U.S. – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that it is highlighting its portfolio of molecular imaging solutions at the Society of Nuclear Medicine and Molecular Imaging (SNMMI) 2018 Annual Meeting in Philadelphia, PA. With innovations such as Time-of-Flight PET and digital PET/CT, Philips is continuing to advance molecular imaging solutions to help seamlessly connect data and technology to enable a better patient experience, confident diagnosis and improved clinical outcomes. “With an increasing number of radiopharmaceuticals entering the market, along with new developments impacting oncology and cardiology, molecular imaging innovation is advancing at an unprecedented rate. As the industry shifts from volume- to value-based care, the need for an accurate treatment pathway is more important than ever,” said Piotr Maniawski, Director of Clinical Science, Nuclear Medicine, Philips. “Philips is making significant strides in nuclear medicine with innovations such as the Vereos Digital PET/CT, which is gaining momentum globally and enabling hospitals to make an immediate impact on outcomes, increasing diagnostic confidence.” During the meeting, Philips was recognized with the “Best of Service” award for PET imaging by IMV ServiceTrak. Philips is committed to providing its customers with service that goes beyond reacting to problems, taking a proactive and preventative approach to drive performance. By combining systems, software and services to deliver integrated, tailored solutions, Philips is helping providers deliver better healthcare through increased staff efficiency and improved patient satisfaction. Integrated solutions drive diagnostic confidence The centerpiece of Philips’ booth at SNMMI will be the Vereos Digital PET/CT situated in an Ambient Experience Suite. Attendees can follow the entire patient journey for a PET/CT scan. Beginning with the pre-scan contrast injections, patients are able to relax in a PET uptake room outfitted with Ambient Experience. They then move to the imaging suite where the Ambient Experience continues, helping reduce patient stress and improve care management. In a study by Philips of 600 imaging patients, respondents identified physician trust, comfort, safety and getting the image right the first time as priorities in their imaging experience [1]. Vereos Digital PET/CT is the world’s first and only fully digital, clinically proven PET/CT solution. It is supported by rigorous clinical evidence, including more than four years of investigational studies and over 100 published clinical studies. Vereos is a well-established, comprehensive solution to help clinicians improve patient care and manage costs. Featuring Philips’ proprietary Digital Photon Counting (DPC) technology, Vereos offers improved detectability and characterization of small lesions [2]. The system provides uncompromised detectability and quantification at half the PET dose [3]. Vereos also provides lesion detectability in one tenth of the time [4]. When used with Ambient Experience, patients can experience a calmer imaging process through soothing audio and visual elements. Philips will also feature Philips CardioMD IV at the show. Specifically designed for nuclear cardiology, this clinically-proven SPECT system is tailored to enhance workflow and add value to radiology departments. With a small footprint, CardioMD IV will fit in virtually any existing camera room without requiring costly renovations. Its patient-friendly design allows for greater positioning flexibility. Half-time cardiac imaging with Astonish reconstruction helps improve workflow efficiency without sacrificing image quality, resulting in cardiac imaging with improved workflow, providing diagnostic confidence and enhanced patient experience. For more information on Philips' molecular imaging solutions and to learn more about the company’s presence at SNMMI 2018, visit booth #617, www.philips.com/SNMMI and follow the conversation on @PhilipsLiveFrom. [1] The Patient Experience in Imaging Study was conducted by Kantar TNS in the Netherlands and commissioned by Philips [2] Hussain T, Nguyen QT. Molecular Imaging for Cancer Diagnosis and Surgery. Advanced drug delivery reviews. 2014;66:90-100. doi:10.1016/j.addr.2013.09.007 [3] Nguyen NC, Image Quality and Diagnostic Performance of a Digital PET Prototype in Patients with Oncologic Diseases: Initial Experience and Comparison with Analog PET, J Nucl Med 2015; 56:1378–1385 [4] Knopp,M, Binzel,K, Bardos,P, Knopp,M, Wright,C, Zhang,J, Nagar,V, Hall,N, Maniawski,P, Next Generation Digital PET/CT: A Phase I Intra-Individual Comparison with Current Photomultiplier TOF PET/CT. Radiological Society of North America 2015 Scientific Assembly and Annual Meeting, November 29 - December 4, 2015, Chicago IL
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and enabling better outcomes across the health continuum from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips' health technology portfolio generated 2017 sales of EUR 17.8 billion and employs approximately 74,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.