Jun 20, 2019
Philips extends advanced automation capabilities on its EPIQ CVx cardiology ultrasound platform, making accurate exams faster and easier to conduct
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced new advanced automation capabilities on its EPIQ CVx and EPIQ CVxi cardiac ultrasound systems. With Release 5.0, both the EPIQ CVx and EPIQ CVxi now include automated applications for 2D assessment of the heart, as well as robust 3D right ventricle volume and ejection fraction measurements, making accurate exams faster and easier to conduct [1]. Together, the new applications provide clinicians with the means to confidently evaluate the heart’s function, increasing diagnostic confidence for patients with pulmonary hypertension, congenital heart disease, coronary disease and heart failure. “The complexity of cardiac exams can be a barrier to accessing high quality care. Philips is addressing this by leveraging Artificial Intelligence to make echo exams easier, faster and more reproducible,” said David Handler, General Manager of Cardiac Ultrasound at Philips. “By incorporating advanced automation, there is less variability between scans, leading to accurate treatment decisions which benefits patients. The new release of EPIQ CVx is a major step forward, reducing the number of touches of the system by 21% in each exam, which is equivalent to more than 400 exams each year [2].”
The new release of EPIQ CVx is a major step forward, reducing the number of touches of the system by 21% in each exam, which is equivalent to more than 400 exams each year.
David Handler
General Manager of Cardiac Ultrasound at Philips
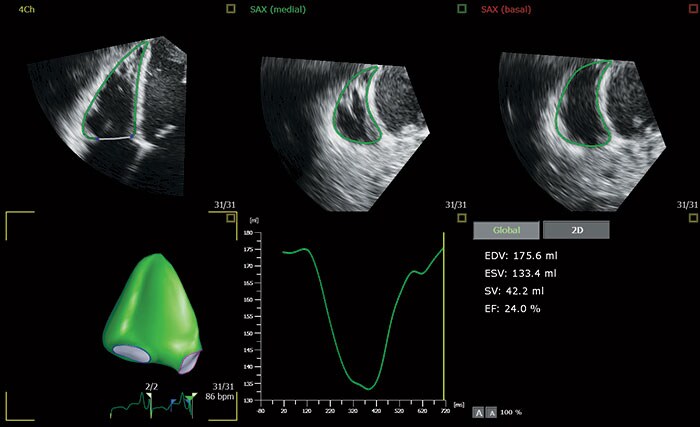
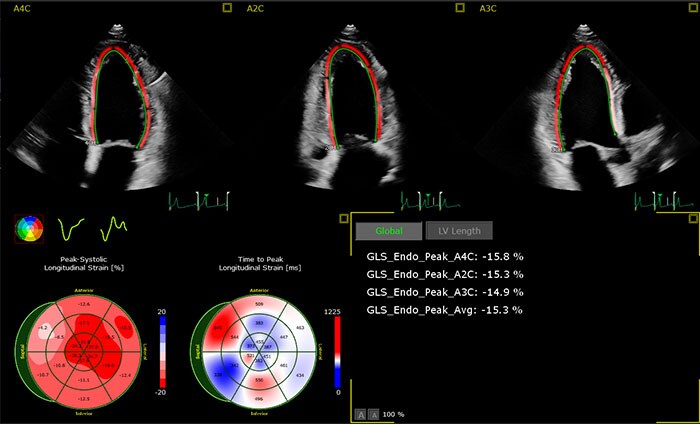
The AutoStrain LV application uses advanced Automatic View Recognition technology to identify the different views of the heart, providing exceptional visualization and analysis of left ventricular function – extremely important diagnostic information for patients at risk of developing cardiovascular disease. Also available are AutoStrain LA and AutoStrain RV, applications which automate the measurement of left atrial and right ventricular longitudinal strain respectively. By creating reliable and reproducible strain measurements for the left ventricle, left atrium and right ventricle, the AutoStrain LV, LA and RV applications support clinicians treating patients with atrial fibrillation, arrhythmia and other complex heart conditions. The 3D Auto RV application segments, identifies the borders of and aligns the views of the right ventricle, enabling clinicians to carry out the quantification and check the measurements in as little as 15 seconds [3]. These new applications expand on the advanced automation applications already available on the EPIQ CVx platform, including Dynamic HeartModel, which provides a clear vision of the heart’s chambers and how well they are pumping blood – specifically on the left side, where heart failure often begins. A recent study of Dynamic HeartModel published in the European Heart Journal – Cardiovascular Imaging concluded that ‘the automated algorithm can quickly measure dynamic left ventricle and left atrial volumes and accurately analyze ejection/filling parameters’ [4]. The new release also adds diagnostic capabilities to the EPIC CVxi. Tailored for use in the interventional lab, the system can now also be used in the echo lab.
Advanced automation makes echo more accessible to a growing patient base
Beyond monitoring overall heart function, new medical fields such as cardio-oncology are using echocardiography to better assess heart health during chemotherapy, which can damage the heart if the dosage is not closely monitored. Cardio-oncology has increased the global demand for echocardiography as clinicians need to perform a deep analysis on cardiac images throughout the patient’s treatment, ensuring measurements are consistent from one exam to the next so that the results can be reliably compared. Philips will unveil EPIQ CVx Release 5.0 alongside its full suite of solutions for diagnosis and interventional guidance during the annual meeting of the American Society of Echo (ASE), taking place June 21-25 in Portland, Oregon. Follow the #ASE2019 conversation on @PhilipsLiveFrom throughout the event and visit Philips on booth #300. More information about the Release 5.0 is available here. [1] The Philips CVx and CVxi 5.0 Diagnostic ultrasound systems are available for sale globally. The AutoStrain LV, LA, RV applications and the Dynamic Heart Model application are CE marked and FDA cleared and available for sale in Europe and in the USA. The AI enabled 3D AutoRV application, designed using a Machine Learning-based algorithm, is CE marked and FDA 510(k) pending, and is therefore available for sale in Europe but is not available for sale in the USA. [2] Based on eight scans per day over 48 weeks. [3] Based on internal test results.
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and enabling better outcomes across the health continuum from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips generated 2018 sales of EUR 18.1 billion and employs approximately 77,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.