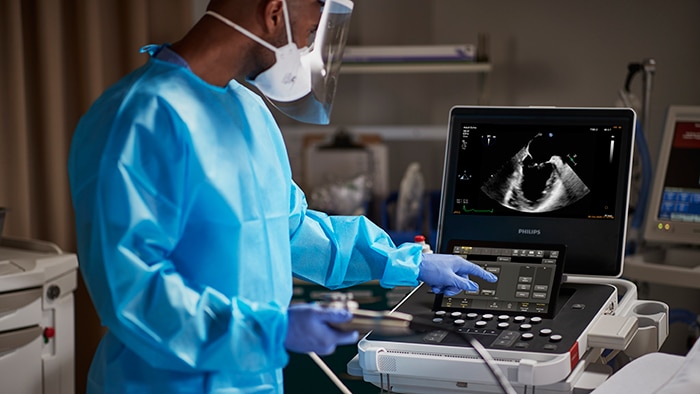
During the American College of Emergency Physicians (ACEP) 2022 Scientific Assembly (Oct. 1 – 4, San Francisco, CA), Philips announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its newest compact ultrasound system, the innovative 5000 Compact Series. This recent clearance elevates ultrasound portability performance through the latest advancements in Philips ultrasound technology designed to deliver cart-based premium image quality in compact form, featuring a wide range of diagnostic ultrasound solutions to fit the needs of many clinical specialties including point of care, cardiology, general imaging, and obstetrics and gynecology.
“Globally, clinicians are faced with increasing pressures to deliver higher patient throughput for quick, confident diagnoses via ultrasound exams, while receiving more and more requests to perform rapid ultrasound exams at the bedside,” said Matthijs Groot Wassink, General Manager of Point-of-Care Ultrasound at Philips. “Until now, portable ultrasound systems have typically meant decreased performance, lack of advanced features, and lower grade image quality. With this clearance for our next generation portable Philips Ultrasound 5000 Compact Series, clinicians now have the ability to make quicker and more confident diagnoses. Patients within locations such as hospitals, surgery centers, clinics, physician offices, and point-of-care settings, also have faster access to quicker results, physician communication and ultimately, increased peace of mind in their treatment plans.”
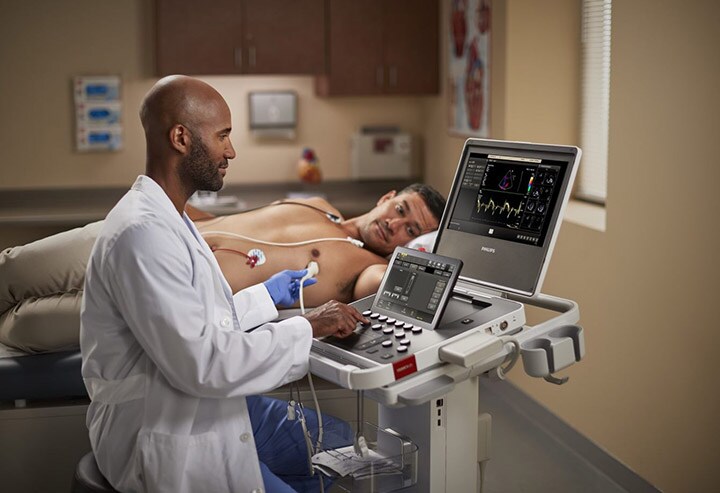
Building on its leadership in ultrasound imaging, Philips pairs portability with performance with this latest innovation. With the new Ultrasound 5000 Compact Series, clinicians have the advantage of a compact system without compromise in performance to deliver clinical decision support with mobile efficiency. Combined with real-time collaboration and remote consultations and training (via Philips Ultrasound Collaboration Live tele-ultrasound capabilities), and integrated connectivity for streamlined workflows, this latest innovation helps clinicians make the right diagnosis the first time.
Offering consistency across Philips ultrasound systems EPIQ and Affiniti, the Philips Ultrasound 5000 Compact Series shares the same user interfaces, features, touchscreens, transducers, and workflows for ease of use and operator efficiency. Designed to provide clear images to quickly inform and to aid diagnosis confirmation, the Ultrasound 5000 Compact Series helps decrease the need for costly, time-consuming repeat scans. With its wide range of clinical capabilities, real-time collaboration, and wireless connectivity with reporting, Philips Ultrasound 5000 Compact Series is one of Philips’ most reliable and robust compact systems.
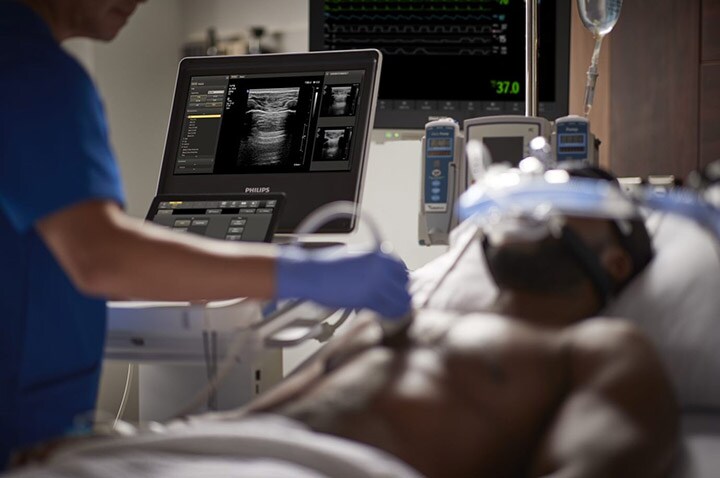
Philips will spotlight its new Ultrasound 5000 Compact Series at the American College of Emergency Physicians (ACEP) Scientific Assembly 2022, Oct. 1-4 in San Francisco. Visitors to the Philips Booth (#1818) at ACEP will see live demonstrations of the latest advanced technology from Philips, including the new Ultrasound 5000 Compact Series, along with demonstrations of Philips Ultrasound Collaboration Live tele-ultrasound capabilities, providing real-time clinician support and remote training for ultrasound exams.
Visit Philips Point of Care Ultrasound for the latest information on Philips point-of-care (POC) solutions for emergency services and follow #ACEP22 for live updates throughout the ACEP event.
Share on social media
Topics
Contact

Kathy O'Reilly
Philips Global Press Office Tel.: +1 978-221-8919
You are about to visit a Philips global content page
Continue