Mar 11, 2022
Philips Collaboration Live integrated tele-ultrasound expands FDA 510(k) clearance for remote diagnostic use to additional mobile platforms
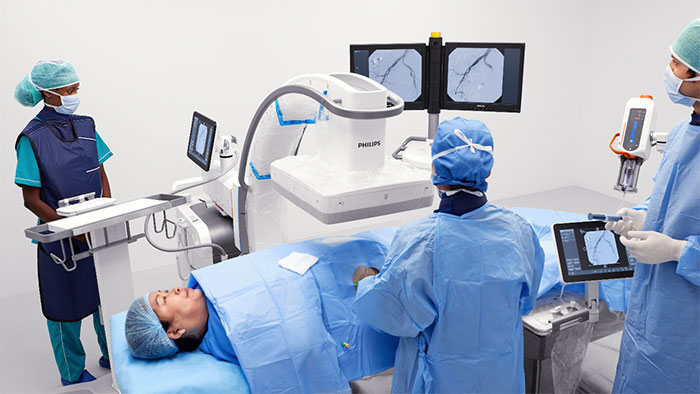
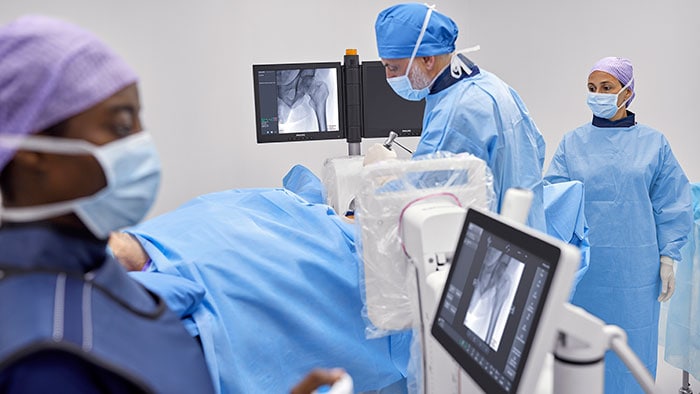
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that Collaboration Live has received expansion of its U.S. Food & Drug Administration 510(k) market clearance for remote diagnostic use on additional mobile platforms. Available on Philips Ultrasound Systems EPIQ and Affiniti [1], Collaboration Live allows clinicians to collaborate in real-time with colleagues in order to complete image acquisition and diagnosis regardless of location. Ultrasound imaging is one of the most accessible and low-cost imaging modalities. However, capturing and interpreting the acquired images and making an accurate diagnosis requires a great deal of expertise and skill. While ultrasound equipment is readily available both in hospitals and healthcare settings, widespread staffing shortages can impede access to care. In addition, the COVID-19 pandemic highlighted the risk to clinicians in contact with infectious patients, and the resulting backlog of postponed exams is now increasing workloads and driving the need for greater efficiency. Collaboration Live enables remote diagnosis, expanding the ability to image patients inside and outside of healthcare facilities, and could help limit the risk of exposure for healthcare workers and patients to infectious diseases such as COVID-19. With its expanded clearance for diagnostic use on additional platforms, clinicians now have the ability to consult or diagnose from their mobile device, check the strength of their bandwidth before making a call, and check their remote screen calibration to ensure image quality is appropriate for diagnosis. This can enable greater efficiency and flexibility for clinicians since they are no longer limited to a PC for remote collaboration. Collaboration Live is already helping to extend access to quality care in underserved settings. "We have been able to open a strictly telemedicine office to serve our patients in rural communities because Collaboration Live enables us to carry out a full diagnostic patient visit remotely,” said Michael S. Ruma, MD, MPH, at Perinatal Associates of New Mexico, who will present peer-reviewed data on the associated return-on-investment at this year’s American Institute of Ultrasound in Medicine Annual Meeting (AIUM 2022).
We are committed to helping our customers deliver better access to care for their patients regardless of their location at any time. Collaboration Live is a perfect example of how Philips is delivering an expanded experience for patients and healthcare professionals through telemedicine.
Gerois Di Marco
General Manager Global Ultrasound Services & Solutions at Philips
"We are committed to helping our customers deliver better access to care for their patients regardless of their location at any time. Collaboration Live is a perfect example of how Philips is delivering an expanded experience for patients and healthcare professionals through telemedicine", said Gerois Di Marco, General Manager Global Ultrasound Services & Solutions at Philips. “Sonographers no longer have to feel they are alone or limited to only acquiring ultrasound images and preparing a report for subsequent clinical review. They can now call on the expertise of colleagues during ultrasound examinations to deliver on-the-spot accurate diagnosis that every patient deserves.” Philips Collaboration Live lets users quickly and securely talk, text, screen-share, and video-stream directly from a Philips Ultrasound System EPIQ or Affiniti to a compatible remote PC or mobile device [1]. In addition to allowing remote clinicians to view real-time diagnostic ultrasound images and communicate with the local clinician and patient to provide guidance and ask questions, the ultrasound user can give remote control [2] of the ultrasound system to the remote user, enabling them to enhance image quality, capture a new image or make a measurement. These combined features as well as the ability to check the calibration of the remote viewing screen provide the high diagnostic confidence needed to help optimize patient outcomes. Collaboration Live can help hospitals standardize care across locations and support remote staff via protocol training, remote support during ultrasound examinations, and real-time communication between clinicians and patients, helping to ensure the same high standard of care wherever it is delivered. Collaboration Live is applicable in any area of medicine – such as breast imaging and hepatology, where ultrasound can provide important diagnostic information – including maternal fetal medicine when dealing with the potential of high-risk pregnancies, as well as cardiac imaging. Visit AIUM 2022 for more detail about Philips participation in the event, and visit Philips advanced tele-ultrasound solutions for more information on Philips Collaboration Live and other solutions in the company’s advanced ultrasound portfolio. [1] Requires an EPIQ or Affiniti ultrasound system with software level 7.0.5. or higher. Mobile access via compatible clients including iOS, Android, Chrome web browser and Windows for diagnostic use requires Collaboration Live 9.0 or higher.
[2] Remote control requires a Windows device. Collaboration Live is available in the USA, and select availability in other countries.
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and well-being, and enabling better outcomes across the health continuum – from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips generated 2021 sales of EUR 17.2 billion and employs approximately 78,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.