Plaque Removal
In-vivo study
Comparison of plaque reduction by Philips Sonicare 1100, 2100, 3100 Series* Power toothbrush versus manual toothbrush
Amini P, Imtiaz U, Li J, Ward M, Nelson M, Ambers J Silverstone Research Group, Las Vegas, NV, USA, 2021

Objective
To compare the reduction in surface plaque by the Philips Sonicare 1100-3100 Series* power toothbrush (PTB) handle, paired with the C1 SimplyClean and C2 Optimal Plaque Control brush heads to manual toothbrush (MTB) following 14 days of home use, and to evaluate the safety of the test products.
Methodology
In this randomized, three-arm, parallel, single- blinded, single-center study, eligible subjects were routine manual toothbrush users aged 18-59, non-smokers, with a minimum of 20 scorable teeth and a minimum plaque score of ³1.8 per Lobene and Soparker Modified Quigley-Hein Plaque Index (MPI) following 3-6 hours of plaque accumulation. Enrolled subjects were randomized to three groups. The PTB+C1 BH group was given a Philips Sonicare 1100-3100 Series* PTB with C1 SimplyClean brush head; the PTB+C2 BH group was given a Philips Sonicare 1100-3100 Series* PTB with C2 Optimal Plaque Control brush head; and the MTB group was given a Colgate Classic Adult MTB. All groups performed toothbrushing twice daily with Crest Cool Mint Gel toothpaste following manufacturer’s instructions. The use of any other oral hygiene products was prohibited during the study period. All subjects received MPI assessment at Baseline, and following two weeks of home use of the study products. Subjects presented to clinic for both visits with 3-6 hours of plaque accumulation. Safety was assessed by intraoral exam and per subject report.
Results
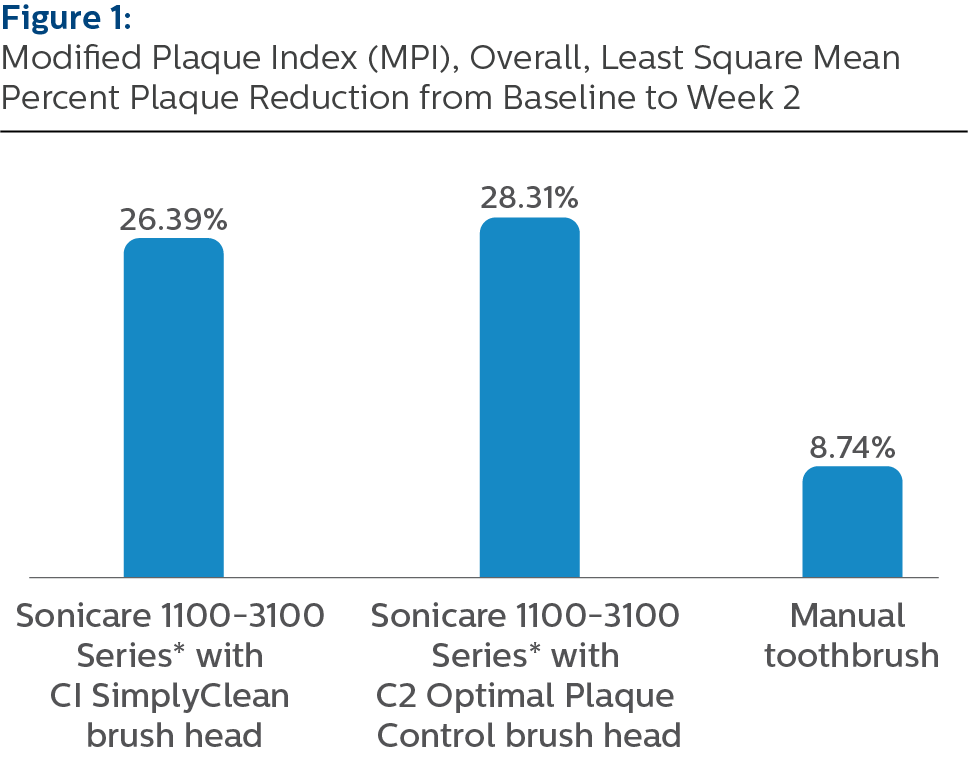
Demographics In total, 240 subjects were enrolled and randomized, and 239 subjects completed the study. There were no statistically significant differences in demographics or baseline characteristics between the three groups. Efficacy Modified Plaque Index (MPI), Least Square Means (SE), Overall
| | PTB+C1 BH | PTB+C2 BH | MTB |
| n | | | |
| Baseline | | | |
| Reduction from Baseline | | | |
| Week 2 | | | |
| Percent reduction from baseline | | | |
| Week 2 | | | |
**Pairwise comparisons were statistically significant versus MTB treatment group, p-value < 0.0001
Safety There were no serious adverse events. One adverse event of mild severity related to the study product occurred in the PTB+C1 BH group.
Conclusions
The Philips Sonicare 1100-3100 Series* powered toothbrush with either C1 SimplyClean brush head or C2 Optimal Plaque Control brush head is statistically superior to manual toothbrush in reducing surface plaque after 14 days of home use. All study products were safe for use.
* Philips Sonicare 1100 Series, 2100 Series, 3100 Series, Sonic Pro 10, Sonic Pro 30 © 2021 Koninklijke Philips N.V. (KPNV ). All rights reserved. PHILIPS and the Philips shield are trademarks of KPNV. SONICARE and the Sonicare logo are trademarks of KPNV.
1 Jan, 2021 by Philips
Reading time: 4-5 minutes
Share on social media
Contacts

Sarah Berryhill
Clinical Marketing Team
You are now exiting the Philips United States (US) site and entering the Philips global site. This content is intended for a global audience. It may not apply to the US and should not be interpreted as meeting US standards, executive orders or regulations.
Continue Assets